Landscape of somatic mutations in ovarian endometriosis and normal uterine endometrium
Clonal expansion and diversification of cancer-associated mutations in endometriosis and normal endometrium
Kazuaki Suda*, Hirofumi Nakaoka*, Kosuke Yoshihara, Tatsuya Ishiguro, Ryo Tamura, Yutaro Mori, Kaoru Yamawaki, Sosuke Adachi, Tomoko Takahashi, Hiroaki Kase, Kenichi Tanaka, Tadashi Yamamoto, Teiichi Motoyama, Ituro Inoue, Takayuki Enomoto
* These authors contributed equally to this work.
Cell Reports DOI:10.1016/j.celrep.2018.07.037
Endometriosis is characterized by ectopic endometrial-like epithelium and stroma, of which molecular characteristics remain to be fully elucidated. We sequenced 107 ovarian endometriotic and 82 normal uterine endometrial epithelium samples isolated by laser-microdissection. In both endometriotic and normal epithelium samples, numerous somatic mutations were identified within genes frequently mutated in endometriosis-associated ovarian cancers. Analyses of mutant allele frequency (MAF), combined with multiregional sequencing, illuminated spatiotemporal evolutions of the endometriosis and uterine endometrium genomes. We sequenced 109 single endometrial glands and found that each gland carried distinct cancer-associated mutations, proving how heterogeneous the genomic architecture of endometrial epithelium is. Remarkable increases in MAF of mutations on cancer-associated genes in endometriotic epithelium suggests that retrograde flow of endometrial cells already harboring cancer-associated mutations have selective advantages at ectopic sites, leading to the development of endometriosis.
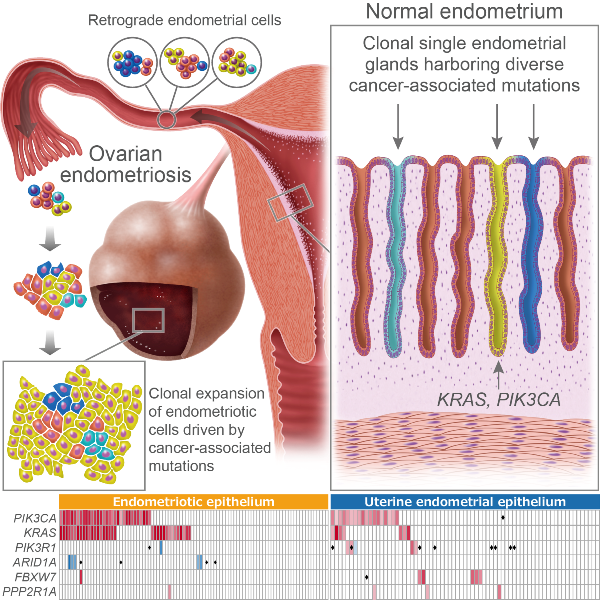
Figure: Graphical representation for retrograde hypothesis of the origin of endometriosis according to the results of this study. Endometriosis and uterine endometrium harbor cancer-associated somatic mutations. Genomic architecture of epithelial cells in uterine endometrium is heterogeneous. Single endometrial glands carry distinct mutations of cancer-associated genes such as KRAS and PIK3CA. Retrograde flow of endometrial cells already harboring cancer-associated mutations have selective advantages and go through clonal expansion at ectopic sites (peritoneal surface and ovary), leading to the development of endometriosis.















