Chemokine signalling controls integrity of radial glial scaffold in developing spinal cord and consequential proper position of boundary cap cells
Division of Brain Function / Hirata Group
Chemokine signalling controls integrity of radial glial scaffold in developing spinal cord and consequential proper position of boundary cap cells
Yan Zhu, Tomoko Matsumoto, Takashi Nagasawa, Fabienne Mackay, Fujio Murakami Journal of Neuroscience 17 June 2015, 35(24): 9211-9224; DOI:10.1523/JNEUROSCI.0156-15.2015Radial glial cells, the CNS neural progenitors, extend long radial processes that form the radial glial scaffold in the developing neuroepithelium. The integrity of this radial glial scaffold is well maintained throughout development. However, how this is achieved while the neuroepithelium rapidly expands and what the consequence might be when this integrity is compromised have not been well understood. In this study, we addressed these questions in the developing mouse spinal cord. We found that CXCR4, a receptor of a chemokine CXCL12 secreted from the pial meninges, is expressed in spinal cord radial glia. Conditional knockout of CXCR4 in radial glia causes disrupted radial glial scaffold with gaps at the pial endfeet layer, and consequentially lead to an invasion of boundary cap cells into the spinal cord. Since boundary cap cells are PNS cells normally positioned at the incoming and outgoing axonal roots, their invasion into the spinal cord suggests a compromised CNS/PNS boundary in the absence of CXCL12/CXCR4 signalling. By interrogating the underlying mechanisms, we then went on to show that CXCL12 signalling promotes the radial glia adhesion to basement membrane components and activates integrin β1 avidity. Our study uncovers an extrinsic molecular regulator for the maintenance of radial glial scaffold integrity during development. And our data suggest that the integrity of radial glia scaffold is important to safeguard the CNS/PNS boundary for the developing spinal cord.
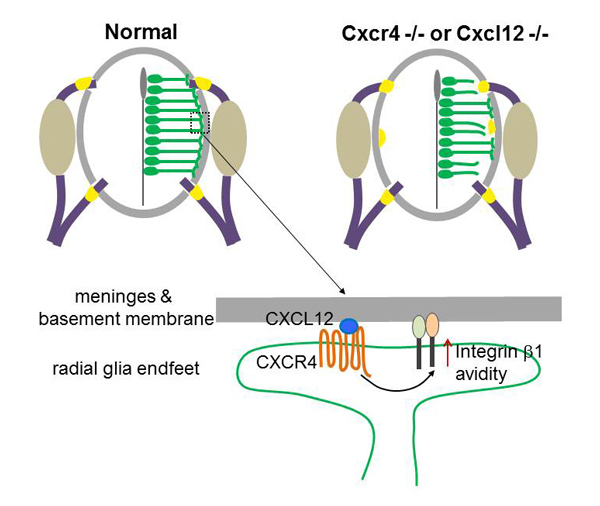
CXCL12 from the meninges regulates adhesion of radial glia to the basement membrane by enhancing avidity of integrin β1. Mice deficient in CXCL12 or its receptor CXCR4 show disrupted radial glial scaffold in their spinal cords, which consequentially causes invasion of boundary cap cells into the spinal cord.















