Development of a CRISPR/Cas9-based tagging method and a degradation inhibitor for the auxin-inducible degron (AID) system
Generation of conditional auxin-inducible degron (AID) cells and tight control of degron-fused proteins using the degradation inhibitor auxinole
Aisha Yesbolatova, Toyoaki Natsume, Ken-ichiro Hayashi, Masato T. Kanemaki
Methods Available online 24 April 2019 DOI:10.1016/j.ymeth.2019.04.010
The auxin-inducible degron (AID) technology developed by the Kanemaki group is based on a plant-specific degradation pathway transplanted to non-plant cells. In human cells expressing a plant E3 ligase component, TIR1, a degron-fused endogenous protein is degraded within 15–45 min upon addition of the phytohormone auxin.
We demonstrated previously the generation of human HCT116 mutants in which the C terminus of endogenous proteins was fused with the degron by CRISPR–Cas9-based knock-in (Natsume et al. Cell Reports, 2016). In this study, A SOKENDAI student, Aisha Yesbolatova, developed new plasmids for N-terminal tagging and described a detailed protocol for the generation of AID mutants of human HCT116 and DLD1 cells (Figure 1).
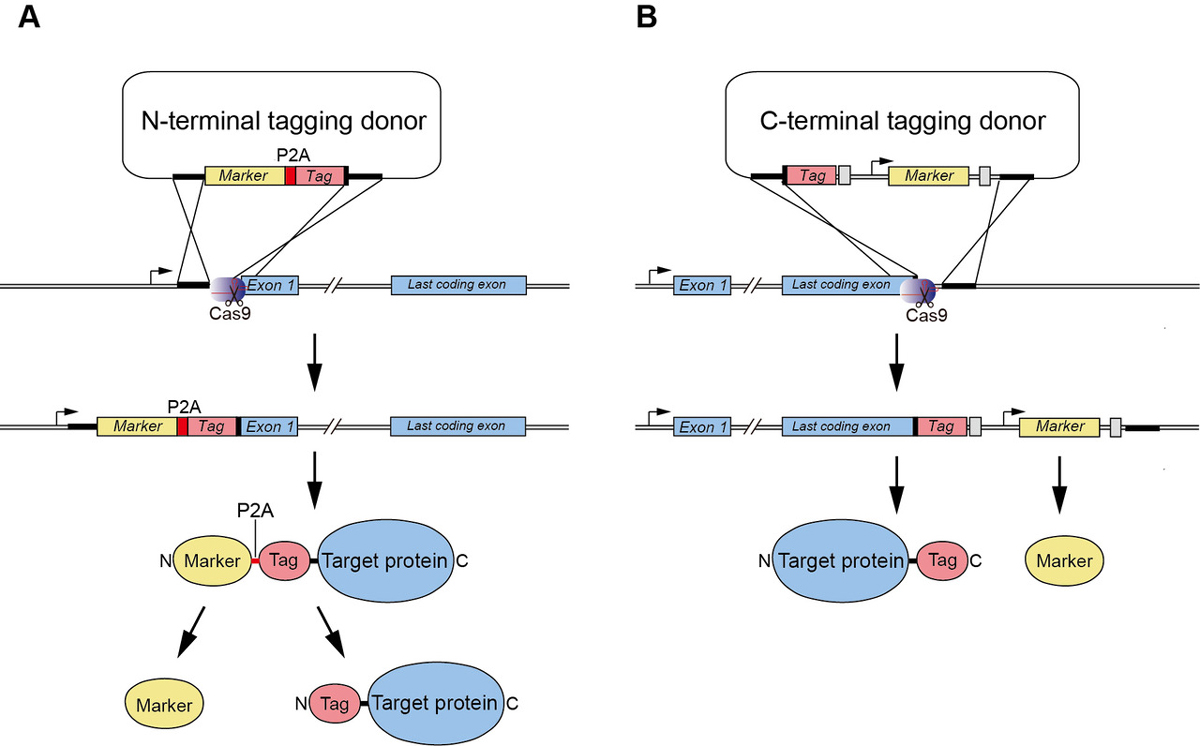
Figure1: (A) A new CRISPR/Cas9-based N-terminal tagging system. (B) C-terminal tagging.
Moreover, we report the use of a TIR1 inhibitor, auxinole, to suppress leaky degradation of degron-fused proteins (Figure 2). The addition of auxinole is also useful for rapid re-expression after depletion of degron-fused proteins.
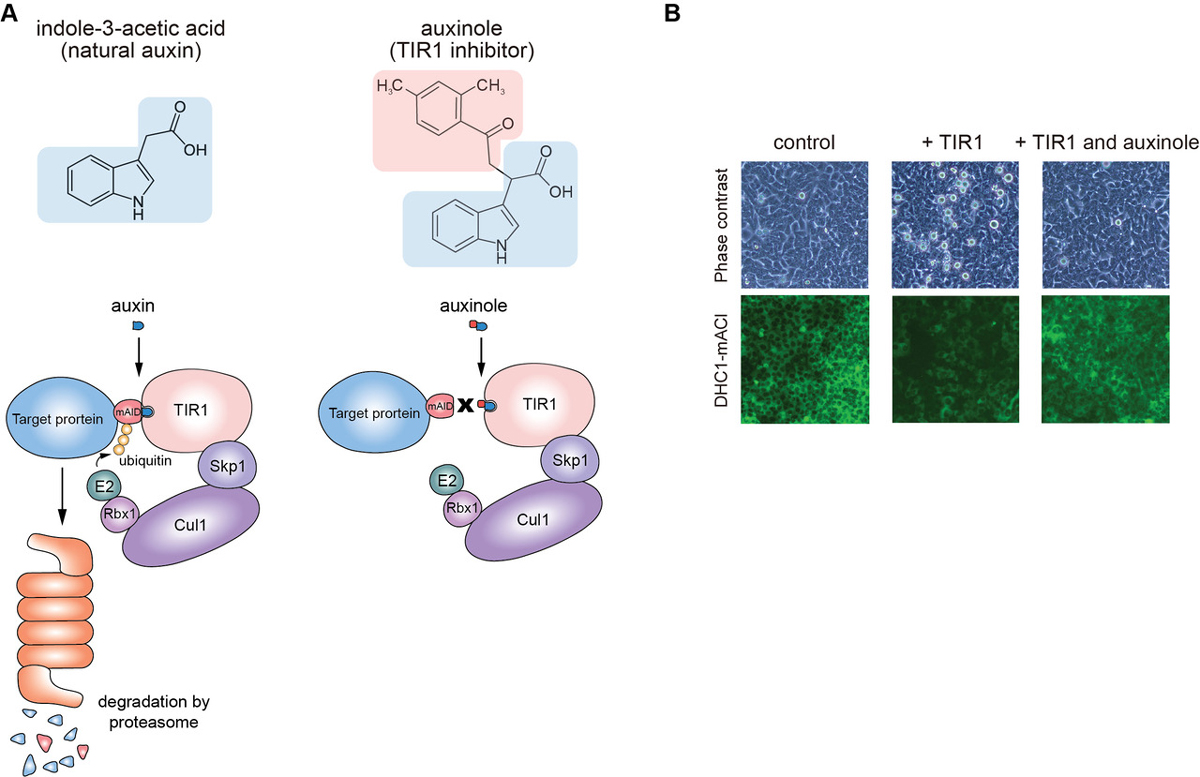
Figure2: (A) The structure of auxin and auxinole, and illustrations showing how these reagents work in the degradation pathway. (B) Suppression of leaky degradation in DHC1-tagged Tet-TIR1 cells. The cells were treated with only doxycycline to induce TIR1 expression or together with auxinole 48 h before microscopy.
We hope that these improvements enhance the utility of the AID technology for studying protein function in living human cells. All described plasmids are available from addgene and RIKEN-BRC.
This study was carried out as a collaboration with Prof. Ken-ichiro Hayashi of Okayama University of Science and was supported by JSPS KAKENHI (17K15068, 18H02170 and 18H04719), JST A-STEP (grant number AS2915150U), the Canon Foundation, the Asahi Glass Foundation, and the Takeda Science Foundation.















