Role and regulatory mechanism of chromatin modifications in gene body
Division of Agricultural Genetics / Kakutani Group
Gene-body chromatin modification dynamics mediate epigenome differentiation in Arabidopsis
Soichi Inagaki, Mayumi Takahashi, Aoi Hosaka, Tasuku Ito, Atsushi Toyoda, Asao Fujiyama, Yoshiaki Tarutani, Tetsuji Kakutani
The EMBO Journal. Published online 18.01.2017 DOI:10.15252/embj.201694983
Heterochromatin is marked by methylation of lysine 9 on histone H3 (H3K9me). A puzzling feature of H3K9me is that this modification localizes not only in promoters but also in internal regions (bodies) of silent transcription units. Despite its prevalence, the biological significance of gene-body H3K9me remains enigmatic. Here we show that H3K9me-associated removal of H3K4 monomethylation (H3K4me1) in gene bodies mediates transcriptional silencing. Mutations in an Arabidopsis H3K9 demethylase gene IBM1 induce ectopic H3K9me2 accumulation in gene bodies, with accompanying severe developmental defects. Through suppressor screening of the ibm1-induced developmental defects, we identified the LDL2 gene, which encodes a homolog of conserved H3K4 demethylases. The ldl2 mutation suppressed the developmental defects, without suppressing the ibm1-induced ectopic H3K9me2. The ectopic H3K9me2 mark directed removal of gene-body H3K4me1 and caused transcriptional repression in an LDL2-dependent manner. Furthermore, mutations of H3K9 methylases increased the level of H3K4me1 in the gene bodies of various transposable elements, and this H3K4me1 increase is a prerequisite for their transcriptional derepression. Our results uncover an unexpected role of gene-body H3K9me2/H3K4me1 dynamics as a mediator of heterochromatin silencing and epigenome differentiation.
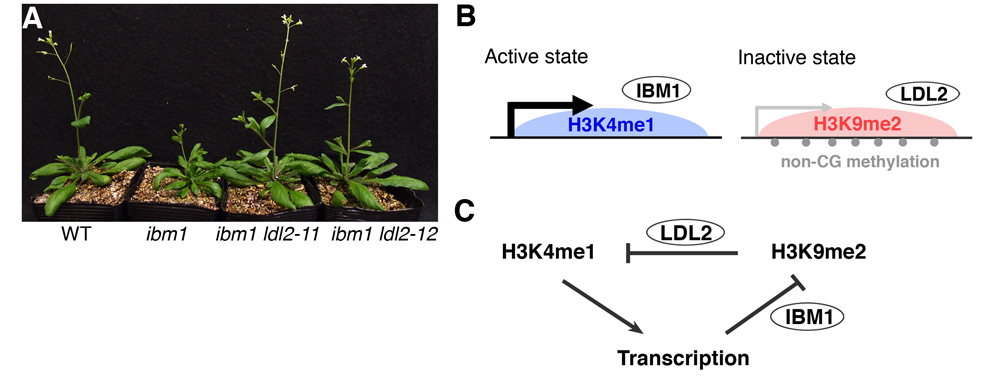
(A) ibm1 ldl2 double mutants show suppression of developmental abnormalities in the ibm1 mutant. (B) Active and inactive chromatin states are determined by two histone demethylases, IBM1 and LDL2. (C) A model for differentiation of bi-stable epigenetic states with feedback regulation. Transcription induces H3K9 demethylation by IBM1 and maintains active chromatin state. Conversely, accumulation of H3K9me2 induces demethylation of H3K4me1 and represses transcription.















