The RNA binding protein Nanos2 organizes a post-transcriptional buffering system to retain primitive states of mouse spermatogonial stem cells
Mammalian Development Laboratory / Saga Group
The RNA binding protein Nanos2 organizes a post-transcriptional buffering system to retain primitive states of mouse spermatogonial stem cells
Zhi Zhou, Takayuki Shirakawa, Kazuyuki Ohbo, Aiko Sada, Wu Quan, Kazuteru Hasegawa, Rie Saba, Yumiko Saga Developmental Cell. Published Online: June 25, 2015 DOI:http://dx.doi.org/10.1016/j.devcel.2015.05.014In many adult tissues, homeostasis relies on self-renewing stem cells that are primed for differentiation. The reconciliation mechanisms of these characteristics remain a fundamental question in stem cell biology. Here, we show that Nanos2, an evolutionarily conserved RNA-binding protein, works with other cellular messenger ribonucleoprotein (mRNP) components to ensure the primitive status of SSCs through a dual mechanism that involves 1) direct recruitment and translational repression of genes that promote spermatogonial differentiation, and 2) repression of the target of rapamycin complex 1 (mTORC1), a well-known negative pathway for SSC self-renewal, by sequestration of the core factor mTOR in mRNPs. This mechanism links mRNA turnover to mTORC1 signaling through Nanos2-containing mRNPs and establishes a post-transcriptional buffering system to facilitate SSC homeostasis in the fluctuating environment within the seminiferous tubule. This research is partly supported by Grant-in-Aid for Scientific Research on Innovative Areas ”Epigenome dynamics and regulation in germ cells” and a “Data assimilation” project of the Transdisciplinary Research Integration Center in ROIS. Zhi Zhou was a NIG postdoctoral fellow 2012.
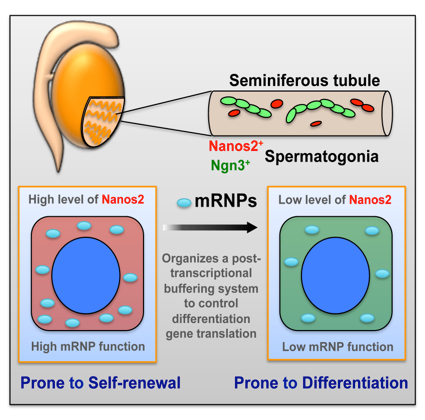
Nanos2 promotes mRNP formation and contribute to the maintenance of undifferentiated state of spermatogonial stem cells. Upon decreasing of Nanos2 level, stem cells shift to differentiation pathway.















