A cell suicide module by use of the auxin-inducible degron (AID) technology
Molecular Function Laboratory • Kanemaki Group
A cell suicide module: Auxin-induced rapid degradation of Inhibitor of Caspase Activated DNase (ICAD) induces apoptotic DNA fragmentation, caspase activation and cell death
Kumiko Samejima, Hiromi Ogawa, Alexander V. Ageichik, Kevin L. Peterson, Scott H. Kaufmann, Masato T. Kanemaki and William C. Earnshaw The Journal of Biological Chemistry, 298, 31617-31623, 2014.; DOI:10.1074/jbc.M114.583542Caspase-activated DNase (CAD), a DNase responsible for DNA fragmentation in apoptosis, is activated upon degradation of its inhibitor ICAD. However, it has not been known whether ICAD degradation is sufficient for induction of apoptosis. To answer this question, we established DT40 cells, in which ICAD can be degraded by addition of auxin using the auxin-inducible degron system. Upon rapid degradation of ICAD, we showed that DT40 cells were induced for apoptosis, indicating that ICDA degradation is sufficient for apoptosis. Moreover, this cell-killing system works in budding yeast cells. We suggest that the cell-killing system we established in this work might be applicable for containment measures of gene-modified organisms in the future.
This study was carried out as collaboration with Dr. Kumiko Samajima and Professor William Earnshaw in University of Edinburgh, UK. Dr. Earnshaw is also an invited professor of NIG.
た。
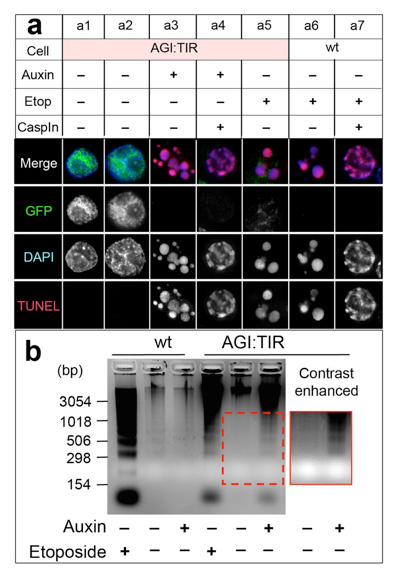
By adding auxin, ICAD was rapidly degraded, resulting induction of apoptosis (a3). In these cells, genomic DNA was fragmented (b).















