DNA binding ability of the cohesin loader stimulates topological DNA entrapment by the cohesin ring
DNA Binding by the Mis4Scc2 Loader Promotes Topological DNA Entrapment by the Cohesin Ring.
Yumiko Kurokawa and Yasuto Murayama.
Cell Reports 33, 108357(2020) DOI:10.1016/j.celrep.2020.108357
The ring-shaped cohesin complex is a member of multi-subunit SMC ATPases, which play vital roles in numerous aspects of chromosome biology including mitotic chromosome segregation, global chromosome organization, transcriptional regulation and DNA repair. Cohesin topologically encircles DNA and is thought to establish series of chromosomal interactions including sister chromatid cohesion by tethering more than one DNA molecule. Using biochemical reconstitution, we show that the ability of the loader to bind DNA plays a critical role in promoting cohesin loading. Two distinct sites within the Mis4Scc2 subunit were found to cooperatively bind DNA. Mis4Scc2 initially forms a tertiary complex with cohesin on DNA and promotes subsequent topological DNA entrapment by cohesin through its DNA binding activity. Furthermore, we show that mutations in the two DNA binding sites of Mis4 impair the chromosomal loading of cohesin. These observations demonstrate the physiological importance of DNA binding by the loader and provide mechanistic insights into the process of topological cohesin loading.
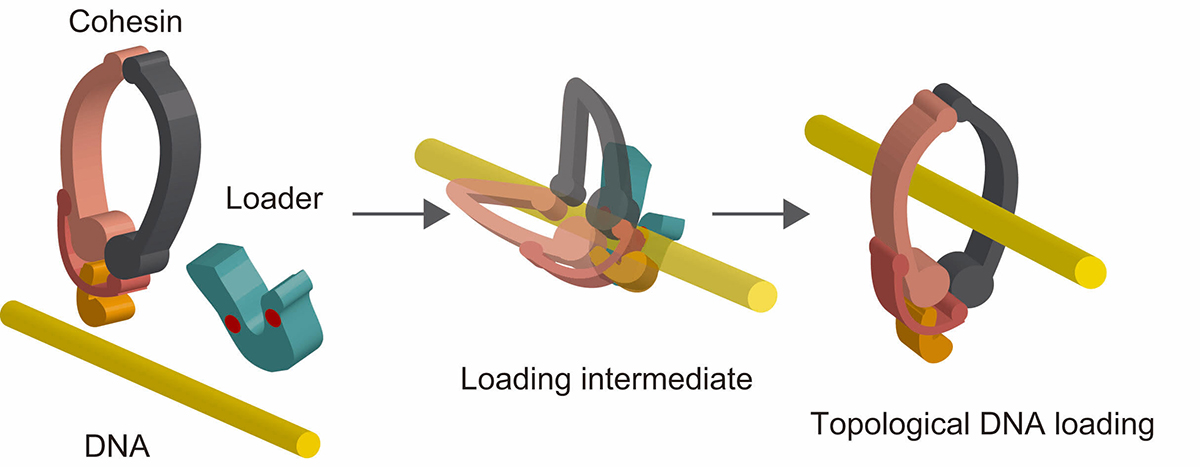
Figure: A model of topological cohesin loading onto DNA mediated by the loader complex. Cohesin forms a tertiary complex with the loader complex on DNA. This stimulates subsequent conformational change of cohesin resulting in topological DNA entrapment.















