Identification of Nanos2 partner protein, Dead end1
Mammalian Development Laboratory / Saga Group
Dead end1 is an essential partner of NANOS2 for selective binding of target RNAs in male germ cell development
Atsushi Suzuki, Yuki Niimi, Kaori Shinmyozu, Zhi Zhou, Makoto Kiso and Yumiko Saga
EMBO report. 2015 DOI:10.15252/embr.201540828
Mammalian Development RNA-binding proteins (RBPs) play important roles for generating various cell types in many developmental processes, including eggs and sperms. Nanos is widely known as an evolutionarily conserved RNA-binding protein implicated in germ cell development. Mouse NANOS2 interacts directly with the CCR4-NOT (CNOT) deadenylase complex, resulting in the suppression of specific RNAs. However, the mechanisms involved in target specificity remain elusive. We show that another RBP, Dead end1 (DND1), directly interacts with NANOS2 to load unique RNAs into the CNOT complex. This interaction is mediated by the zinc finger domain of NANOS2, which is essential for its association with target RNAs. In addition, the conditional deletion of DND1 causes the disruption of male germ cell differentiation similar to that observed in Nanos2-KO mice. Thus, DND1 is an essential partner for NANOS2 that leads to the degradation of specific RNAs. This research is partly supported by Grant-in-Aid for Scientific Research on Innovative Areas ”Epigenome dynamics and regulation in germ cells” to YS and “Mechanism regulating gamete formation” to AS.
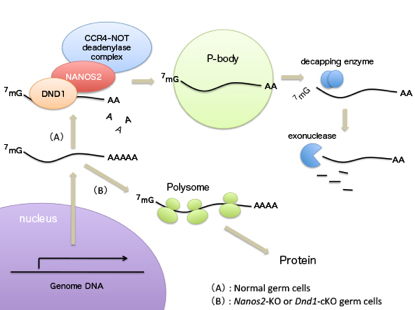
Nanos2 directly interacts with DND1, by which target RNAs are recruited to P-body for degradation. This mechanism is essential for male germ cell development.















