


Visual stimuli entering the retina are converted into neural signals and sent to the brain. The retina converts the elements of the color, shape, and motion of visual objects into separate signals. Professor Yonehara has made a breakthrough in this research field by elucidating the mechanism that detects the direction of movement of an object.
Profile
2003 Graduated with a degree in Veterinary Medicine, Faculty of Agriculture at the University of Tokyo. 2008 Received his PhD from the Graduate University for Advanced Studies/National Institute for Basic Biology, then began working as a postdoctoral fellow in the same laboratory. 2009 Postdoctoral fellow at the Friedrich Miescher Institute for Biomedical Research, Switzerland. 2015 Associate Professor and Nordic EMBL Group Leader, DANDRITE Institute, Department of Biomedicine, Aarhus University, Denmark. October 2021 – present Professor at the National Institute of Genetics
Not the start I had imagined

The origin of my research goes back to my graduate school days. It all started with a discovery in the neural circuits of the mouse retina.
In 2003, after completing my course in veterinary medicine at the University of Tokyo, I joined Professor Masaharu Noda’s laboratory at the National Institute for Basic Biology (NIBB) as a graduate student. As a book lover, I read in Professor Susumu Tonegawa’s “Spirit and Matter” (Bunshun Bunko, 1993) that he traveled all over Japan to look for a graduate school, and I also visited a number of laboratories and put a lot of energy into choosing a school. The laboratory I chose, the Noda Laboratory, was perfect for me because I wanted to elucidate neural circuits based on the experimental techniques of molecular biology.
I was in high spirits before I began, but unfortunately, I was faced with a lot of hardships one after another. I was studying a group of genes that work in the neural circuits extending from the retina to the brain, but I could not obtain the data I wanted. The days went on and on like that. I even thought of giving up on being a researcher many times.
But then, a stroke of luck came my way. A gene in mice that I was analyzing for other purposes led to an important discovery.
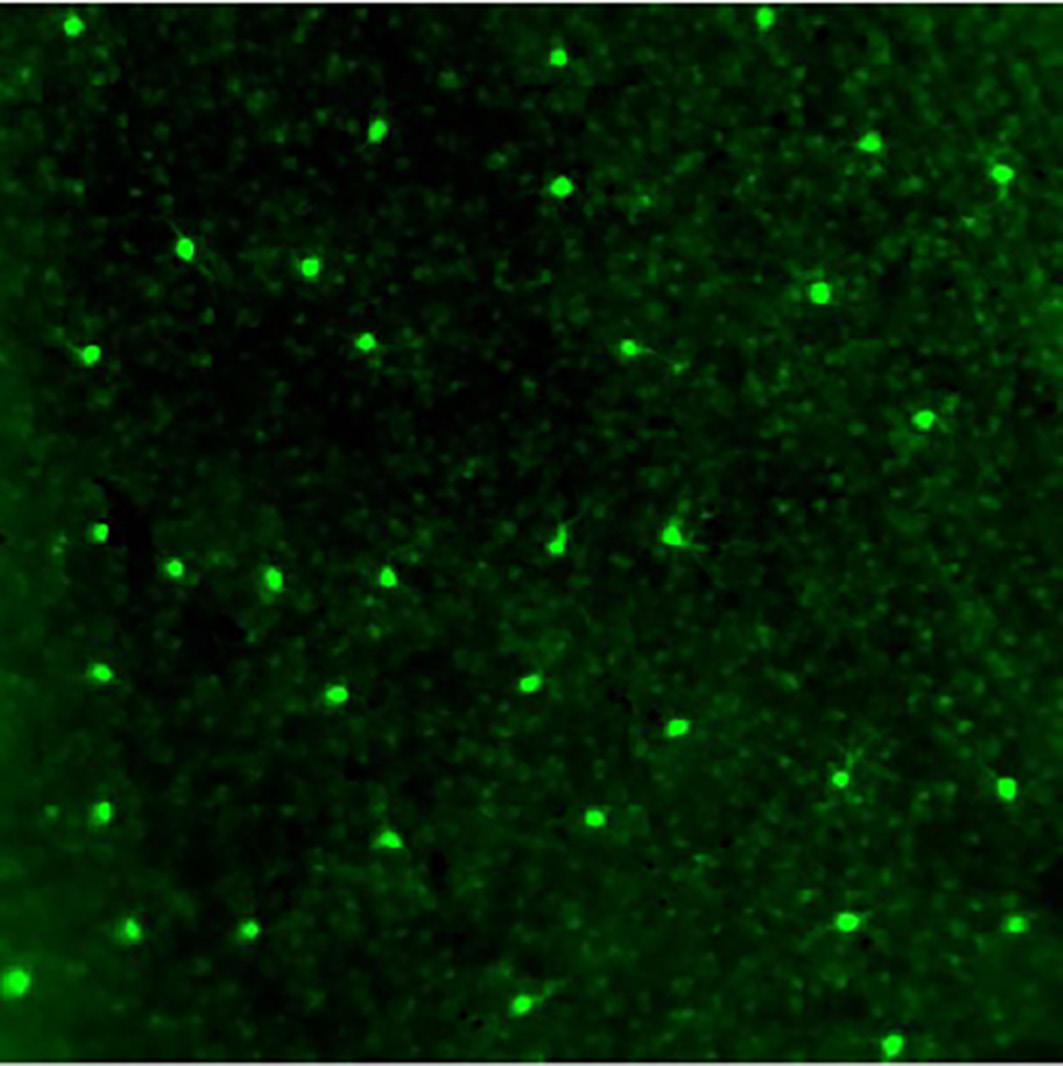
We were studying the expression pattern of a gene called SPIG1 in the retina. The gene had been modified with green fluorescent protein (GFP) so that the resulting protein would fluoresce when expressed. While looking at a microscopic image of the retina, I noticed that the fluorescence of the GFP labeling appeared in a mosaic-like pattern. They were present as spots on the retina, equally spaced on cells that were far apart from each other. Thinking that this GFP labeling might capture some important features of the cells in the retina, I quickly made a decision to change the topic of my doctoral thesis to the analysis of these cells.
Labeling direction-selective cells

The retina of the eye is a light-sensitive organ. Visual stimuli received there are transmitted from the retina to the brain. I looked at where the fluorescent GFP-labeled cells in the retina extended to the brain. It appeared that they extended to a region called the medial terminal nucleus (MTN) of the accessory optic system (AOS).
A review of past literature on the MTN of the AOS indicates that it is involved in sensing direction selectivity (in the upward and downward direction) of visual signals.
So what is “direction selectivity?” Before we discuss it, I will explain the characteristics of the visual circuitry of the retina. It is known that visual stimuli received by the retina are extracted into component signals, such as color, shape, distance, the direction of movement, and speed of the object being viewed, and sent to the brain. This means that vision selectively detects and perceives information that is important to the organism, rather than just vaguely perceiving the entire landscape. So, how is it possible to detect that an object in the field of vision is moving? It has long been known that there are cells in the retina that selectively respond to motion in a certain direction and send signals to the brain. Suppose we consider four directions of motion: up, down, left, and right. It means that there are cells that respond selectively only when the object moves upward, downward, to the right, or to the left. This property is called “direction selectivity.”
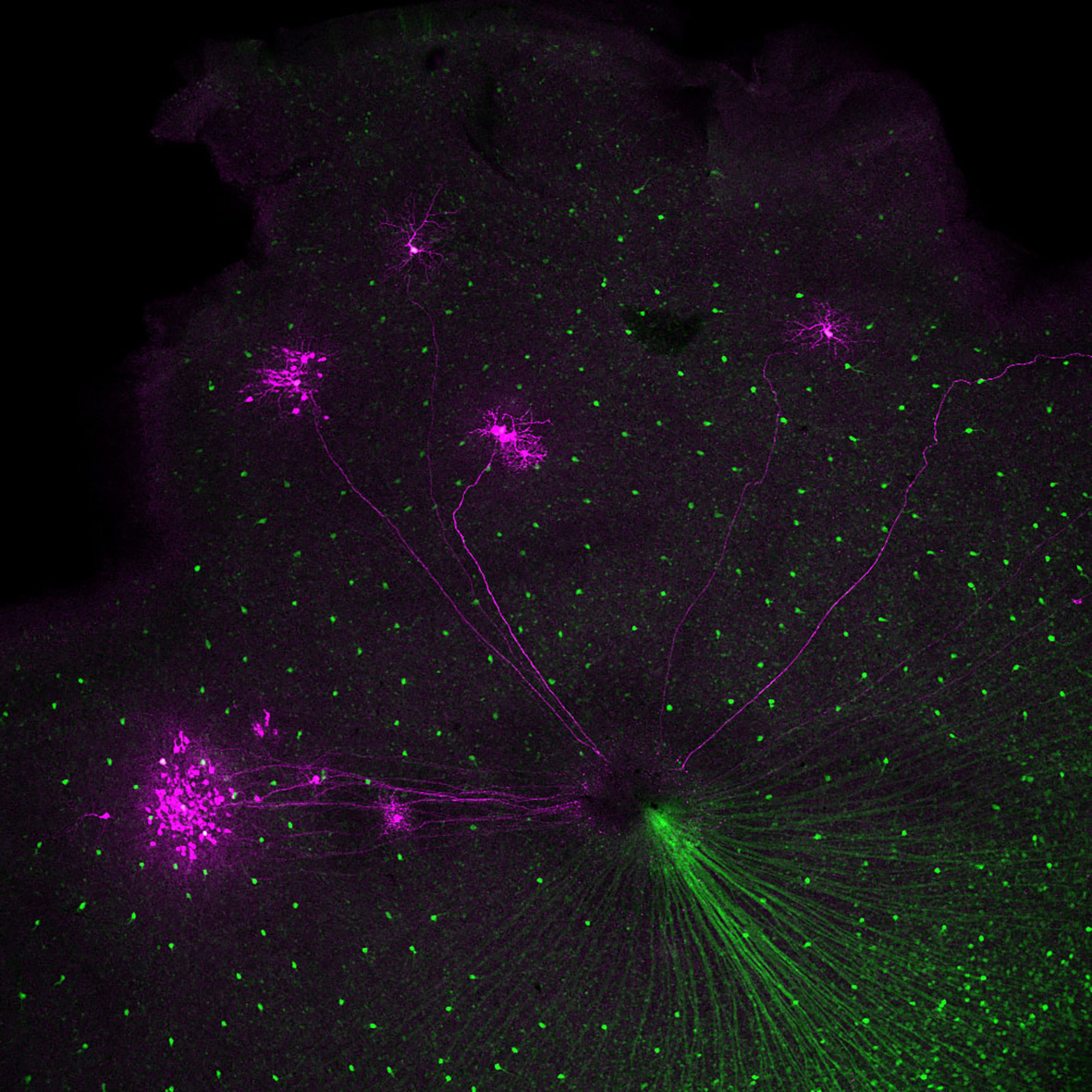
I thought that the GFP-labeled cells in the retina might be direction-selective cells, and to confirm this, I traced the neural pathway from the brain to the retina, this time in the reverse direction. To do this, I used a staining technique called retrograde tracing. As a result, we were able to confirm that the medial nucleus does indeed lead to ganglion cells in the retina.
Here is what was very surprising. Half of the cells traced from the brain to the retina matched the GFP-labeled cells. However, the other half were not GFP-labeled cells. This means that the circuit to the medial nucleus of the brain extended not only from GFP-labeled cells but also from non-GFP-labeled cells. So, GFP-labeled cells might have either upward selectivity or a downward selectivity, and non-GFP-labeled cells have the opposite selectivity. In other words, the GFP labeling should identify cells with either one of these types of direction selectivity. When I realized this, I began trembling at the thought that this would be an important discovery. I still vividly remember the moment when I looked into the confocal microscope in the darkroom and jumped up in surprise.
That day became the starting point of my research that continues to this day. Until that point, no method had been discovered to experimentally investigate the direction selectivity of retinal cells. It was impossible to select cells with direction selectivity from the vast number of cells that make up the retina and record their responses. However, with the use of GFP-labeled cells, this is now possible.
Quickly drafting a thesis ahead of a fast-approaching doctoral defense
In order to prove that my findings were valid, it was necessary to conduct further experiments using electrophysiological techniques. However, since electrophysiology was not available in the Noda laboratory, we applied for a joint research project with Dr. Hiroshi Ishikane of RIKEN (currently Professor at Senshu University) to investigate the actual response of GFP-labeled cells to the direction of motion of visual stimuli in mice.
Meanwhile, I was nearing the end of my doctorate. With the tight timeline for my degree, I did not have much time to finish this research as a dissertation. So, with a little sadness, I decided to present my dissertation in two parts. First, without waiting for the results of the electrophysiological experiments, I summarized the previous results of the discovery of direction-selective cells in a paper. This was published in PLoS One in 2008 (1). Later, the results of the electrophysiology experiments were in line with our expectations and we were able to demonstrate that GFP-labeled cells are direction-selective and that they respond only to upward movement. This paper was published in PLoS One in 2009 (2).
The response to these two papers was very strong, as evidenced by the response to our poster presentation at a conference of the Society for Neuroscience. They were also selected as “must-read” papers by the international paper evaluation system F1000.
I believe the significance of this research result is twofold. One is that we can now analyze direction selectivity by labeling cells with GFP using the SPIG1 gene. Since this gene is expressed in mice from the embryo stage onwards, we now have a means of studying how direction selectivity develops in the retina. F1000 praised this research as “opening up a new field of genetic analysis of retinal neurons.”
The other significant point is that we now know that this upward direction selectivity is genetically determined in cells. There are many examples of cells in the nervous system acquiring specific properties through experience. It had been thought that cells with direction selectivity could also be of that type, but our study demonstrates that this is not the case.
Saved by books


I learned the importance of one’s attitude towards research when I was a postgraduate student at Noda Lab. That was probably the most important thing I acquired during my postgraduate years. Initially, I tended to stick to my own ideas and did not listen to other opinions very much. However, in the Noda Lab, I realized that communication and being open-minded are very important in research, and presumably in any job.
As I said earlier, when I was young, apart from research and sleeping, all I did was reading books. I often sought an answer in books when distressed or faced with a dilemma. In real life, you are surrounded by only a limited number of people, but you are able to access the experiences of so many authors through their books. It would be a loss not to learn from the enriching writings of our wise predecessors.
For example, during my postgraduate years, Peter Drucker’s notion that ‘practice will lead to competence’ had a strong influence on me. Until I learned this quote, I thought that a person’s competence was determined by nature and education, and that the only thing remaining was to acquire knowledge. But when I encountered the quote, I decided to change my attitude. Since then, I have tried to determine how I should act and how I respond by looking at matters more objectively.
“Conversations with Goethe”( Iwanami Bunko, published 1968-69)left the greatest impression on me when I was a postdoctoral fellow. It was written about how to be first class – if you would like to create something new, then you must learn from your predecessors. There is nothing new but only in the accumulation of the past. Do not become self-complacent when seeking originality – these quotes became guidelines for my actions.
Continuing research in the Roska lab
After completing my PhD, I wanted to further develop my analysis or direction selectivity in the retina. I did some research on where to apply as a postdoctoral researcher, and the Botond Roska lab in Switzerland seemed like the best place to start. The Roska lab was located at the Friedrich Miescher Institute for Biomedical Research in Basel at the time (he later founded and became director of the Institute for Molecular and Clinical Ophthalmology in Basel). Dr. Roska was among the first to document the optical response of the mammalian retina using two-photon excitation (TPE) microscopy. The laboratory had the equipment to conduct TPE microscopy and electrophysiological analyses important for studying neural circuits in the retina, as well as viral vectors and even optogenetics, which was still a rarity at the time.
I immediately sent an application letter to Dr. Roska with an idea for a study using GFP-labeled cells. I was invited for an interview and duly hired. Professor Noda then graciously allowed me to bring my GFP-labeled mice with me.
On the day I arrived at the Roska lab, I had a six-hour discussion with Dr. Roska and we decided on several research themes. What is the mechanism by which cells acquire the direction selectivity signal? Previous research had shown that the signals (input signals) that ganglion cells, which make up the retina, receive from surrounding cells are spatially asymmetric. This asymmetry was hypothesized to be the mechanism that causes direction selectivity. In a letter to Dr. Roska, I explained my idea that we could demonstrate the mechanism of direction selectivity acquisition by studying how asymmetric input signals develop in mice using GFP cells. That became the first research topic we would undertake. Dr. Roska gave me the idea for the specific experimental methods to study this. This method used optogenetics, electrophysiology, and TPE microscopy.
I learned these techniques so that I could use them in my own experiments, and set to work. In the retina, there are cells called “starburst amacrine cells” that send the received visual stimuli to ganglion cells. When we examined the input from these cells to GFP-labeled cells, we found that up to the sixth day of life, signals are input to visual stimuli from any direction, but in the second week of life, signals are input only to visual stimuli from a fixed direction (upward). In other words, the study revealed the development of spatial asymmetry in signals and proved that it confers direction selectivity on ganglion cells in the retina. The results of the study were published in Nature in 2011 (*3). I felt that this was the end of the first stage of my research that had begun with papers published in PLoS One in 2008 and 2009.
研究結果は、2011年にNatureに掲載されました(*3)。2008年と2009年のPLoS One掲載論文で始まった一連の研究が、これで一段落したという思いでした。
Some of the research from my time in the Roska lab was published in Neuron in 2016 (*4), which many people consider to be the culmination of my research as a whole. The following study identified FRMD7 as the gene essential for spatial asymmetry.
We knew early on that although the SPIG1 gene used for GFP labeling could identify direction-selective cells, it was unrelated to the cause of direction selectivity (and spatial asymmetry of the signal). So the race was on to find out what genes were essential for this mechanism.
I discovered the FRMD7 gene by studying a genetic disease. When our eyes look out the window on a moving train, for example, they follow the flowing scenery. This is a natural reflex of the eye moving to stabilize the visual field, but it is known that this reflexive movement does not occur in people with a genetic disease called congenital nystagmus. Seventy percent of people with this genetic disease have a deficiency in the FRMD7 gene, which causes the disease.
One day, I was looking at a database of experimental results from the Roska lab. I noticed that the starburst amacrine cells in the retina expressed this FRMD7 gene. Perhaps congenital nystagmus was a disorder of direction-selective cells in the retina. When I mentioned this to Dr. Roska, he was excited, and we went directly to a mouse breeding facility and arranged to create knockout mice lacking a functional FRMD7 gene. Analysis of these mice revealed that they lacked direction selectivity to recognize horizontal movement, and we were able to identify FRMD7 as the cause of this defect.
The discovery of this gene had another major significance. Previously, it had been thought that only lower mammals and other vertebrates had direction selectivity in retinal cells. However, my research provided the first strong evidence that this was not the case and that it might also be present in the human retina. Since then, there has been a lot of work on the analysis of direction selectivity in primate retinas.
Starting up an independent laboratory


I was able to write many good papers in the Roska lab and learned a lot from Dr. Roska’s vision for his research. I thought that the next step was to finally have my own lab, so I started preparing and sent my application for a position to about 50 places around the world.
I was able to write many good papers in the Roska lab and learned a lot from Dr. Roska’s vision for his research. I thought that the next step was to finally have my own lab, so I started preparing and sent my application for a position to about 50 places around the world.
In 2015, I arrived at the DANDRITE Institute at Aarhus University as Associate Professor and Nordic EMBL Group Leader. TPE microscopes use a mechanism that allows fluorescent dyes to be illuminated by low-energy near-infrared light, so the field of view is not too bright and experiments can be performed to record how labeled cells respond to the light. We were able to have this microscope in our own laboratory, and nothing could have made me happier. With our own microscope, we were able to modify the system, tweaking the wavelength of the light stimulus, for example, to make it suitable for us.
Some of our research results at Aarhus University have not yet been released, but I will talk about a few that have already been published. Direction-selective cells, the type that send signals to the AOS, tend to respond strongly when the speed of the object being viewed is slow and less strongly when the speed is fast. This speed dependence is important for controlling the reflex movements of the eye for visual field stabilization, and we have elucidated its mechanism (*5).
There is also a type of cell in the retina called the “retinal bipolar cell.” It was not well understood whether these cells emit direction-selective signals. When I was in the Roska laboratory, we also conducted research in this area and concluded that they did not emit such signals, however, our analysis here has produced experimental results that overturned our previous conclusion (6). The response of each individual synapse was measured with a TPE microscope, and a large amount of data was analyzed using machine learning. We believe that the results were obtained through more precise analysis. In addition to this, we also identified the region of the cerebral cortex that signals from the retina reach by analyzing FRMD7 knockout mice (7).
Winning a grant in Europe

When I started up my own lab at Aarhus University, I obtained research grants in addition to the start-up funding available from the university. I will explain about research grants in Europe, including the ones I got during my post-doctorate.
There are four types of fellowship that are important when doing research as a postdoc in Europe: the Human Frontier Science Program Long-Term Fellowship, the Marie Skłodowska-Curie Individual Fellowship, the EMBO Long-Term Fellowship, and the Japan Society for the Promotion of Science Postdoctoral Fellowship for Research Abroad. Of these, I was selected for the EMBO fellowship and the JSPS overseas fellowship programs.
For independent researchers, there is also the European Research Council (ERC), a large research funding agency in Europe. The ERC has grants for different stages of a researcher’s career and is a very competitive grant. I was fortunate to be selected for the Starting Grant by the ERC. When I started up my lab at Aarhus University, I was able to use the combined funding from this grant and the Aarhus University startup budget, which was very helpful.
You can apply for an ERC research grant after you have been selected for a position at a university where you will start your own company. The review process, including interviews, took about a year. In writing the application, I first collected three or four applications from past successful grant recipients to learn how they wrote their applications. Then, I asked a number of people, Nobel laureates among them, to read and critique my application in order to improve it. Once you pass the review process, the next step is an interview. After a 15-minute presentation, there is a 5-minute question and answer session. Of course, I had been diligently practicing for the interview.
The time I put the most energy into practicing interviews was when I was trying to get a position as an independent researcher after the Roska lab. In European universities, I think most interviews take the form of a symposium where about six candidates are assembled and present for about 30 minutes in a symposium format. Every day, I practiced my presentation over and over again, and afterwards, I repeated the process of presenting in front of everyone and making corrections in response to criticism. I also videotaped my presentations and reviewed them to check my pronunciation and body language. In addition to Aarhus University, I had interviews at several other universities. The more I practiced, the clearer it was that I was getting better at what I was doing.
Research expanding to the National Institute of Genetics


I arrived at the National Institute of Genetics (NIG) in October 2021 and have a dual appointment with Aarhus University until 2023. I was able to obtain several grants in Japan too, which enabled me to purchase two new TPE microscopes. One of the things we hope to do at NIG is analyze primates. I would like to start working with marmosets and see how much of what we found in mice is also shared with primates, or how different they are. I would also like to link this to findings that will be useful for understanding human vision and treating diseases.
In addition, I use mice in my research, and the NIG has a large-scale mouse breeding facility. In addition to the excellent breeding environment, we have a collection of mouse subspecies from all over the world. I would like to use these mice to begin research on the mechanisms of vision and environmental adaptation. What I mean is that mice have a habit of fleeing when they see their natural enemies, such as hawks and owls, flying overhead. Experiments have confirmed that when a black circular paper is placed over the mice, they flee to the shelter. We would like to investigate from the genetic level how this response is affected by differences in the habitat of each subspecies of mice, whether there are differences in natural enemies, and whether there is a relationship with the shape of birds. I have high expectations that our research will expand from its focus on the retina to higher level vision systems as a whole.


What I expect from students is …well, they don’t have to understand what’s interesting yet, but I want to recruit candidates who are motivated to achieve something at a high level and who enjoy developing themselves. As for postdoctoral fellows, I prefer people who have a clear mindset of what their goals are.
In my youth, I worked hard without breaks since I was well-trained in laboratories in Japan. Initially, at the Roska Lab, I continued to work outrageously hard and eventually I became unwell. At that point, I realized that my health is more important than anything else. Since then, I try to spend more time with my family and to relax by taking holidays and going traveling. Looking around the European colleagues around me, I was so amazed to see that even those with outstanding achievements could all afford to enjoy their research. Therefore, I emphasize the importance of all my lab members enjoying their research. That is the best way to progress and develop my research on neural circuits in the retina in the future.
Interviewer: Yoshiko Fujikawa, Science writer
Photographer: Mitsuhiko Kurusu, NIG ORD
August 2022
References
- *1 Expression of SPIG1 reveals development of a retinal ganglion cell subtype projecting to the medial terminal nucleus in the mouse
PLoS One. 2008 Feb 6;3(2):e1533. - *2 Identification of retinal ganglion cells and their projections involved in central transmission of information about upward and downward image motion
PLoS One. 2009 Jan 29;4(1):e4320. - *3 Spatially asymmetric reorganization of inhibition establishes a motion-sensitive circuit
Nature. 2011 Jan 20;469(7330):407-410. - *4 Congenital Nystagmus Gene FRMD7 Is Necessary for Establishing a Neuronal Circuit Asymmetry for Direction Selectivity
Neuron. 2016 Jan 6;89(1):177-93. - *5 Spatiotemporally Asymmetric Excitation Supports Mammalian Retinal Motion Sensitivity
Curr Biol. 2019 Oct 7;29(19):3277-3288. - *6 Direction selectivity in retinal bipolar cell axon terminals
Neuron. 2021 Sep 15;109(18):2928-2942.e8.. - *7 A segregated cortical stream for retinal direction selectivity
Nat Commun. 2020 Feb 11;11(1):831.
